Gold nanoclusters for cancer nanomedicine
Gold nanoclusters can deliver drugs precisely to cancer cells without damaging healthy tissues. Using atomistic molecular dynamics simulations, researchers at the University of Jyväskylä Nanoscience Centre showed that the ratio of cancer-sensing and drug molecules is a critical parameter in optimizing the properties of gold clusters to maximize the probability of cluster adhesion to the surface of cancer cells.
The computation of the study was performed on the CSC’s supercomputer Mahti with Grand Challenge resources awarded in January 2021 and the University of Jyväskylä’s FCCI cluster (Finnish Computing Competence Infrastructure).
Gastric (stomach) cancer (GC) is one of the most aggressive cancers with a low early diagnosis rate. Current GC therapies are focused on conventional chemotherapy, where chemo drugs act as a potent non-specific intracellular poison to inhibit cell division. They can kill the cancer cells, but at the same time, they are toxic for healthy tissues. Therefore, there is an urgent need for developing more effective treatment methods for GC to overcome its increasing incidence.
In this sense, water-soluble and atomically precise gold nanoclusters (AuNCs) have emerged as a promising material for cancer nanomedicine due to their exceptional features, such as excellent biocompatibility, superior stability, and efficient renal clearance.
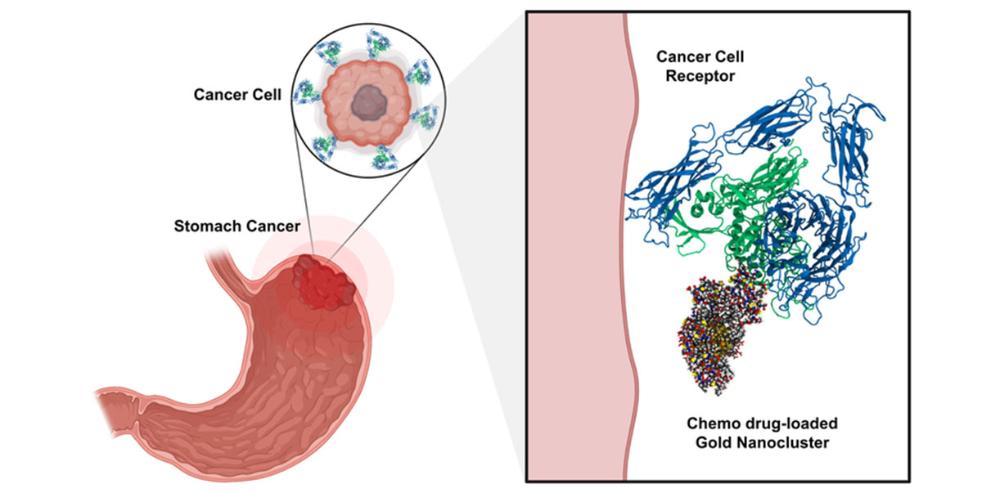
After the simulations, the research continues with laboratory experiments. Francisca Matus, lead author of an article published on the study, will use the data from the calculation to develop suitable peptide molecules for targeting action and chemo drug molecules. The results of the simulations will allow researchers to reduce the number of experiments needed.
– These computational methods provide efficient screening tools to design optimal nanosystems and allow selecting the best candidates among many options which optimizes resources in the experimental stage that follows, says postdoctoral researcher Francisca Matus who is the lead author of the study.
This research extends the Nanoscience Centre’s gold research into the field of nanomedicine.
– This exciting work expands our knowledge and internal collaborations at Nanoscience Center of the University of Jyväskyla to nanomedicine, comments Professor Hannu Häkkinen, who coordinated the study.
More information
Ligand Ratio Plays a Critical Role in the Design of Optimal Multifunctional Gold Nanoclusters for Targeted Gastric Cancer Therapy. María Francisca Matus, Sami Malola, and Hannu Häkkinen.
ACS Nanoscience Au 2021. DOI: 10.1021/acsnanoscienceau.1c00008
Writer: Tommi Kutilainen
